Chronic wounds are those that do not heal in the expected time frame after receiving sufficient treatment. Although there are many intrinsic factors that prevent healing, the most prominent complications are often bacterial infection, biofilm formation, and pathological inflammation. In recent years, excellent work has been done to document and investigate the microbiomes of these wounds [1-3]. Inspired by this work, we took on a new research focus for our lab to study the wound microbiome.
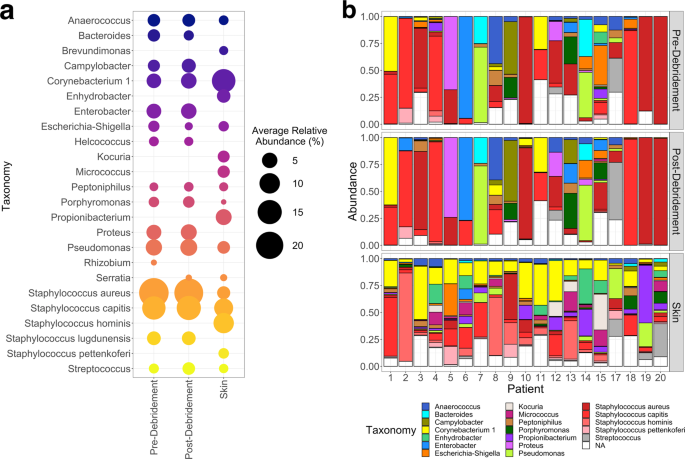
Our initial challenge was study design. We needed to find a balance between statistical power, affordability, and feasibility, for a kind of study where we had little prior expertise. We settled on a cohort of 20 chronic wound patients with single-timepoint sampling from the wound (before and after debridement) and healthy skin. Not ideal, but sufficient to address our hypotheses in our first clinical foray. After a crash course in high-throughput library preparation, sequencing, and 16S rRNA bioinformatics, we had some basic community composition and diversity results that aligned with previous results in the literature.
Still, we wanted to identify specific bacteria that were associated with skin or wounds, debridement state, and healing outcomes. We utilized an RNA-Seq tool (DESeq2) for differential abundance analysis, but recognized that our work would benefit from some statistical expertise. Enter Professor Juhee Lee from UCSC, who built, validated, and applied a novel Bayesian association model for this study. Unsurprisingly, both methods concordantly associated pathogens with wounds, and commensals with skin. However, associations to pre- and post-debridement samples were limited to low-abundance taxa; the application of a second technique allowed us to zero in on the few associations to healing outcomes that were consistent between the two methods.
We were also interested in the oxygen requirements of the microbiomes, since the wound is a dynamic environment with variable oxygen tension in the wound bed, oxygen gradients across the biofilm, and because many wound treatments oxygenate the wound (e.g., debridement, hyperbaric oxygen treatment). We found that wounds that healed were slightly associated with anaerobic bacteria, while nonhealing wounds were strongly associated with facultative anaerobes and slightly with aerobes. It is tempting to speculate that the strict anaerobes are more easily cleared as the wound heals and oxygen tension increases, and that facultative anaerobes are resilient and persistent in the ever-changing wound environment. Still, the prevalence of facultative anaerobes may be correlated to a different, unidentified wound feature that causes poor healing. These findings present intriguing opportunities for future work on causal relationships.
Performing a new clinical investigation is daunting for any research group, but arguably even more so when it is your lab's first clinical investigation, with a brand new graduate student working on an unestablished project. Simultaneously, it can be the most exciting (and overwhelming) undertaking for a PI and their student. Every step is a first - from the initial IRB application to the final statistical analyses. Yet each of these new experiences provides a wealth of knowledge that build the foundation of future work in the group. And so we forge ahead.
[1] M. Loesche, S. E. Gardner, L. Kalan, J. Horwinski, Q. Zheng, B. P. Hodkinson, A. S. Tyldsley, C. L. Franciscus, S. L. Hillis, S. Mehta, D. J. Margolis, and E. A. Grice, Temporal stability in chronic wound microbiota is associated with poor healing, J Invest Dermatol 137 (2017), no. 1 237–244.
[2] S. E. Gardner, S. L. Hillis, K. Heilmann, J. A. Segre, and E. A. Grice, The neuropathic diabetic foot ulcer microbiome is associated with clinical factors, Diabetes 62 (2013), no. 3 923–930.
[3] L. R. Kalan, J. S. Meisel, M. A. Loesche, J. Horwinski, I. Soaita, X. Chen, A. Uberoi, S. E. Gardner, and E. A. Grice, Strain- and species-level variation in the microbiome of diabetic wounds is associated with clinical outcomes and therapeutic efficacy, Cell Host Microbe 25 (2019), no. 5 641–655 e5.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in