The rapid international transmission and emergence of SARS-CoV-2 variants poses a serious challenge to global health, and has rendered our current antibody and vaccine strategies less effective or ineffective. Like other coronaviruses, SARS-CoV-2 utilizes the RBD of envelope homotrimeric spike glycoprotein to interact with cellular receptor ACE-2. Neutralizing antibodies (nAbs) that effectively block RBD-ACE2 interaction represent potential prophylactic and therapeutic potentials and provide guidance for vaccine design. We and others have isolated a large number of nAbs against SARS-CoV-2 from convalescent individuals and provided important insights into the antibody response during natural SARS-CoV-2 infection. As complexity associated with genetic background and sophisticated antibody mutation processes, it is expected that antibody response elicited by SARS-CoV-2 infection varied among different individuals.
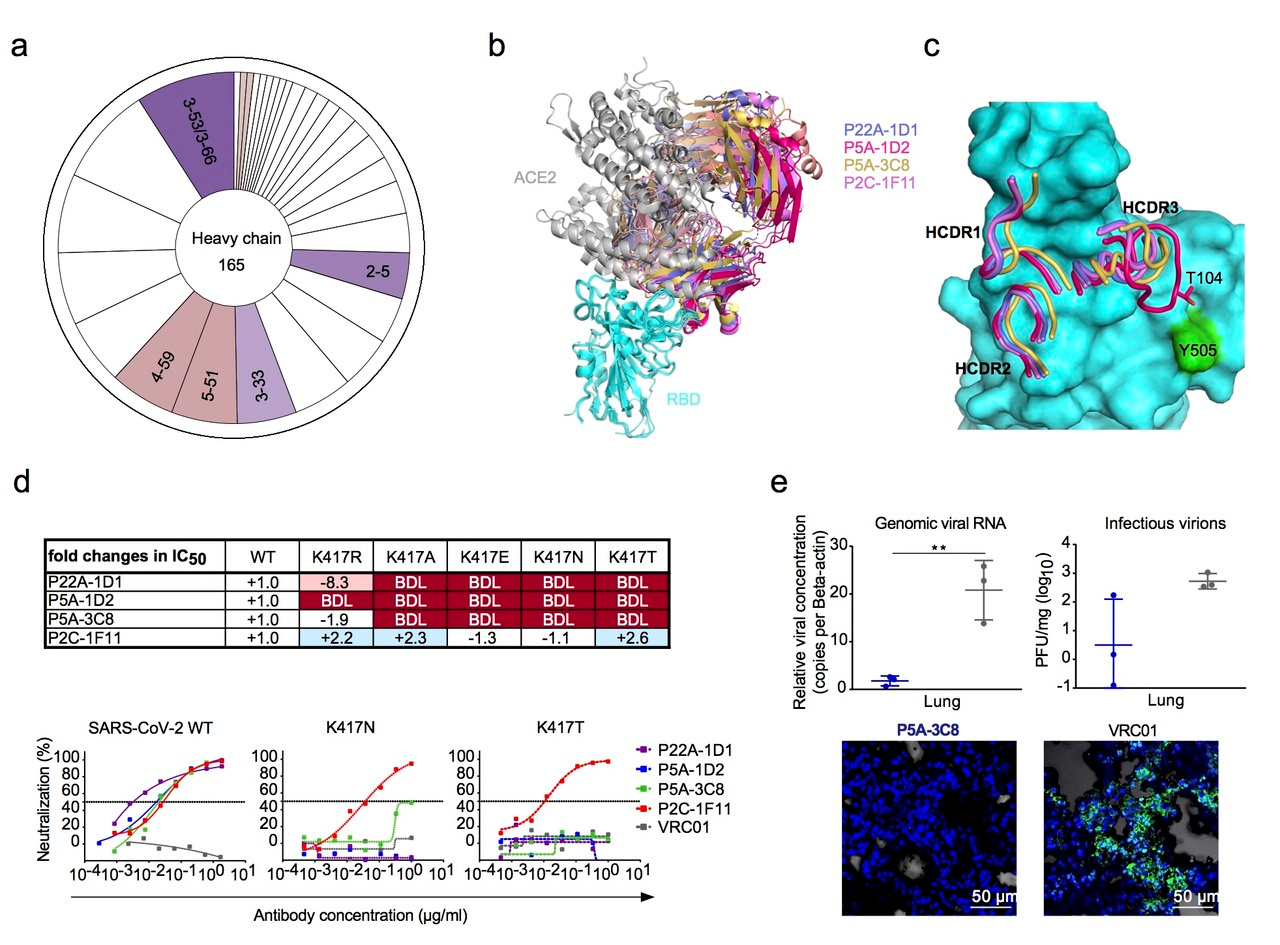
However, recent studies have identified pattern of convergence in antibody lineages cross different COVID-19 patients. Particularly, a disproportionally higher number of nAbs was found from several common genetic families. Of which, the most dominant was derived from IGHV3-53/3-66 family (differed in only one amino acid in framework region 1), suggesting that different individuals could share some public patterns and pathways in antibody response during SARS-CoV-2 infection. Similar findings have also been previously reported for individuals infected by dengue, HIV-1, with influenza vaccination or in other immune settings. Such commonality offers important insights to better understand of human humoral response and provides guidance for our future design of vaccines in capitalizing these unique features.
Here, we interrogated a total of 165 antibodies isolated from eight infected COVID-19 patients in our previous study. Crystal structures and mutagenesis analysis revealed the existence of public antibodies shared among 3 of the 8 patients. These antibodies shared IGHV3-53/3-66 in their heavy chain and characterized by their high neutralizing potency to SARS-CoV-2, similar angle of approaching to RBD, competitive capacity and binding footprints with ACE2 on RBD. One of their representatives, P5A-3C8, demonstrated highly protective efficacy in a golden Syrian hamster model against SARS-CoV-2 infection. However, single mutation at position 417 emerged during in vitro selection was found to confer resistance to these antibodies. We further investigated the impact of all naturally occurring mutations at position 417 on binding and neutralization of public antibodies, which have recently been identified in SA501Y.V2 (B.1.351) variant from South Africa and in BR501Y.V3 (P.1) from Brazil, and found capable of substantially reducing antibody and vaccine efficacy including those already approved for emergence use. These findings highlight the intricate and evolving interaction between SARS-CoV-2 and antibody response, indicating our immune response is one of the important drivers in selecting escape variant during natural infection.
Taken together, the identification and characterization of public antibodies and their escape variants help us to better understand the interplay between antibody response and viral evolution, and to provide insights for next generation antibody cocktail and vaccine design.
Figure 1. a. Preferred usage of IGHV3-53/3-66 among the potent neutralizing antibodies. b. Crystal structures of RBD and Fab complexes. c. Conserved HCDR1, HCDR2, and different HCDR3. d. Susceptibility of K417 variants to neutralization of public antibodies. e. Efficacy of P5A-3C8 prophylaxis against live SARS-CoV-2 infection in Syrian hamsters.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in